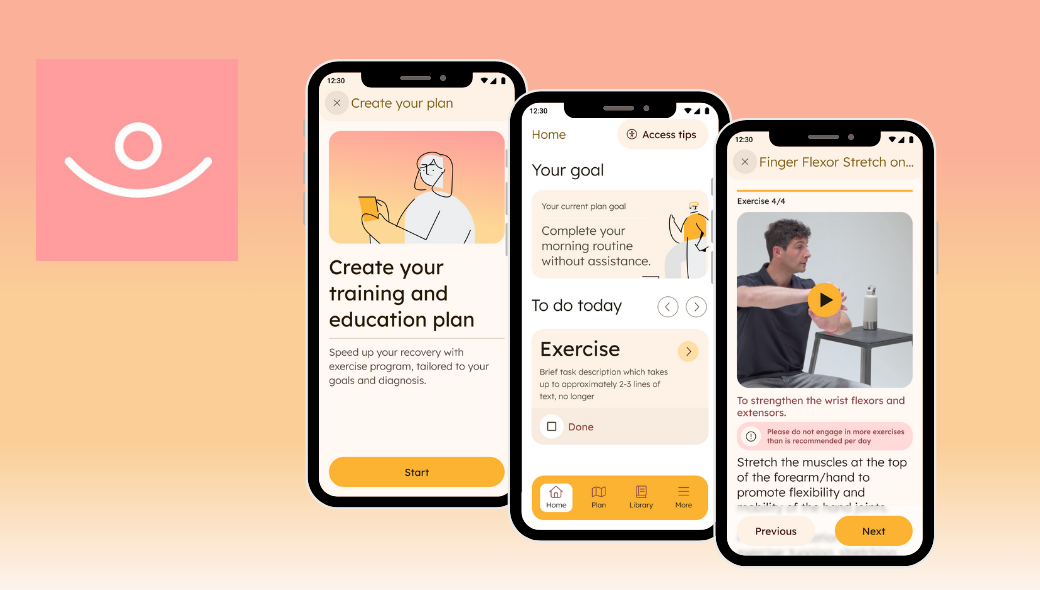
At S3 Connected Health, we create digital health solutions and digital therapeutics (DTx) that improve the lives of people with acute and chronic conditions.
We support our partners in pharma through co-creation, development, and commercialization of solutions, bringing them to market quickly, operating them efficiently, and ensuring successful adoption.





























-1.png)



-1.png)

.png)

.png)