Case Study
TrackSMA: a digital health solution for rare disease
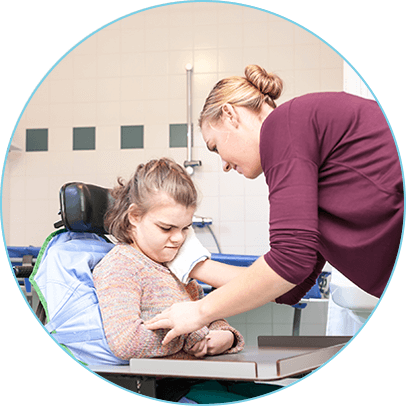
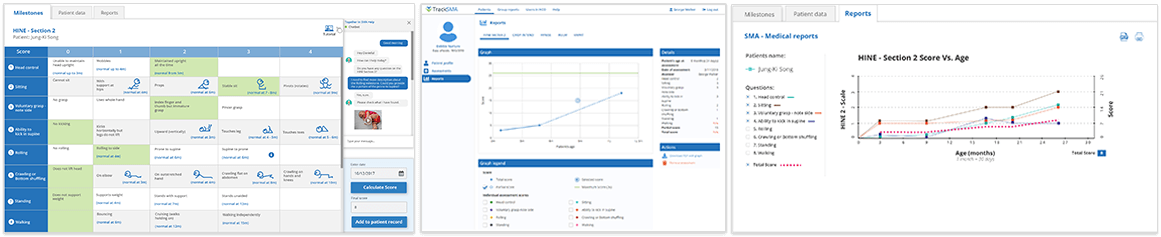
TrackSMA is a digital health solution that facilitates the capture of validated clinical assessments for spinal muscular atrophy (SMA).
The solution was developed by S3 Connected Health partnering with pharma leader, Biogen. It is currently deployed in the APAC region where it enables clinical teams to record and visualize patient progress and disease progression.
TrackSMA drives better insights on the management of patients and supports care teams to make more informed decisions on the treatment of patients.