As the digital health industry has grown, so has the complexity of digital health tools and the range of tasks they can fulfill.
This offers those who choose to implement digital health solutions the chance to build more effective, far-reaching tools that drive real impacts for patients; and for patients themselves, it means better, more personalized, continuous care.
For pharma leaders just starting out in digital health, however, picking the right solution can be daunting.
To help choose the best solution in which to invest, we’ve outlined the three key categories of digital health solutions that pharma might use, as well as what they entail, how they interlink with each other, why pharma might choose one over the others, and the implications this will have for the development process.
Evaluating your options
There are three main categories for pharma to consider
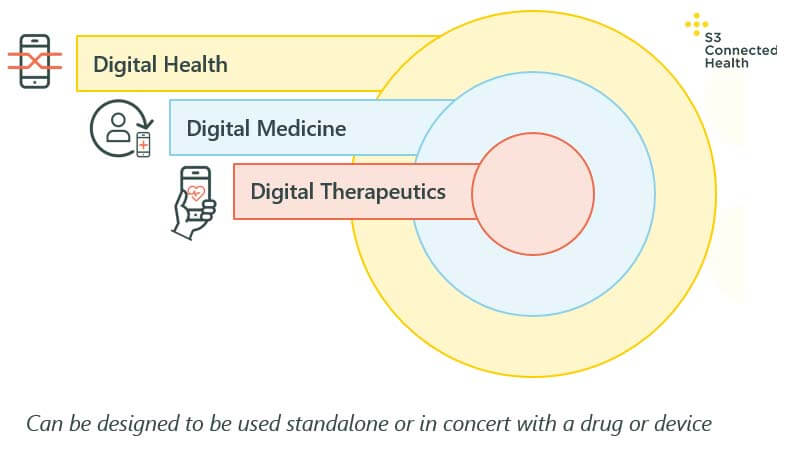
It’s important to note that the particular digital health category pharma selects will be determined by the intended use of that product. This, in turn, will determine the regulatory requirements of a product, the evidential requirements, and its path to market.
With that in mind, there are three main categories to consider, as you can see in the diagram above:
-
Digital health is the overarching term for digital technologies in the health and wellness space. Some are regulated, some aren’t, depending on the product’s intended use.
-
Digital medicine is a subset of digital health, used to describe tools that support the practice of medicine, including tools for measurement, tools for treatment, recovery, prevention, and health promotion.
-
Digital therapeutics (DTx) is a further subset of digital medicine: DTx are digital solutions that deliver medical interventions directly to patients using evidence-based, clinically evaluated software to treat, manage, and prevent a broad spectrum of diseases and disorders. Typically, DTx will require significant evidence that shows the treatment is efficacious, as with a drug or other medical device.
No matter which category a solution falls into, it can operate as a standalone tool, or be used in conjunction with drugs and/or medical devices.
Choosing the right solution for pharma
Drivers for pharma companies in digital health
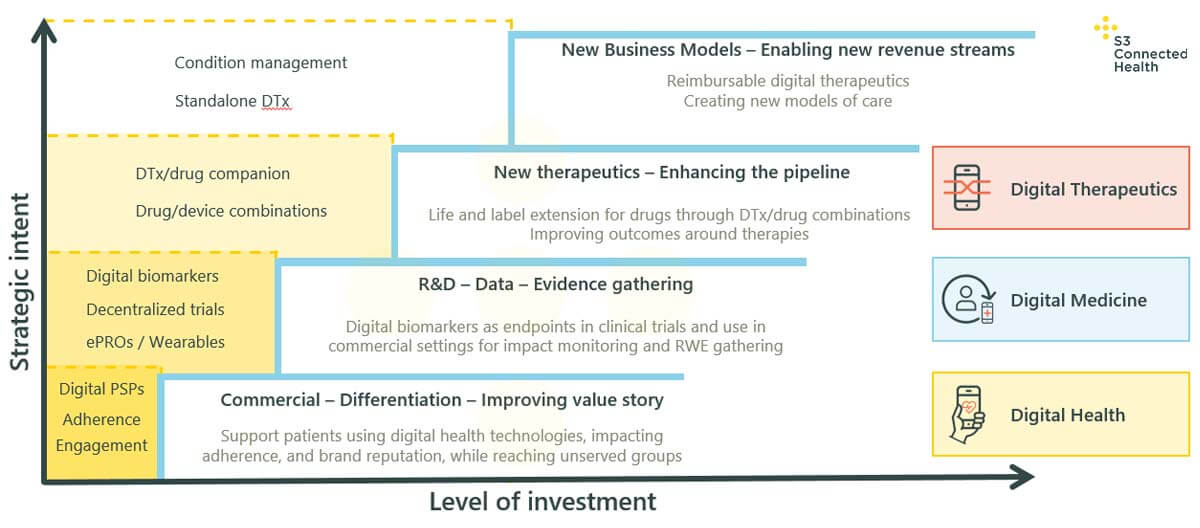
How, then, can pharma companies know the level of solution they need?
A good place to start is identifying the business objectives driving the need for a solution. These could come from anywhere across the business, from franchise and brand teams, to R&D, medical affairs, innovation teams, or, increasingly, from a broader, purpose-built digital health function or department.
In the graph above, the central section shows the key drivers for each respective category:
-
Digital health: In terms of objectives fulfilled, where the driver is improving performance or differentiating a pharma brand by offering digital solution(s) that support patients with adherence, or supporting patients already on therapy with other challenges related to their condition, this falls into the broadest category of digital health. The need for solutions here is typically driven by brand or franchise teams.
-
Digital medicine: For R&D teams looking to identify new digital biomarkers, or create decentralized trials, this will fall into the category of digital medicine. A key driver of these solutions is the need for evidence and data. Both goals will often entail connected wearable solutions, the gathering of ePRO data, and will help connect patients to clinicians for support.
-
Digital therapeutics: Where the key driver is to respond to a need to improve patient outcomes or extend the drug pipeline through life and label extensions for drugs or new digital/drug combinations, solutions will fall into the category of digital therapeutics. The resulting solutions could be combined apps, dosage calculators, or a combination of a digital therapeutic with a drug.
As an extension of this section, where the objective is new revenue models, this is often fulfilled by standalone DTx or condition management solutions that demonstrate evidence of improvements in outcomes, and are reimbursable.
Whatever the key objective or goal driving the development of a particular digital health solution, pharma needs to understand that the higher you go up – that is, the more complex the solution you choose – the greater the investment required, and the greater the burden of evidence you’ll need to provide, for both regulators and payors to support reimbursement. Additionally, the level of internal stakeholders that need to be involved in the project will also increase, as more complex projects will typically take longer, cost more money, and entail more risk.
A clear goal will smooth your product development pathway
While the range of digital health solutions on offer may seem complex, it doesn’t have to be. It’s simply a question of understanding the aims and objectives you’re looking to achieve, as well as what each type of tool requires to be a real success, from stakeholder involvement, to evidential requirement, costs, and development time.
Rest assured, you don’t have to undertake all of these decisions alone. You can find out more about the partnership models to help you create the best possible digital health and DTx partnership models in our recent blogs.