Case Study
Rare Diseases Patient Support Program across multiple territories

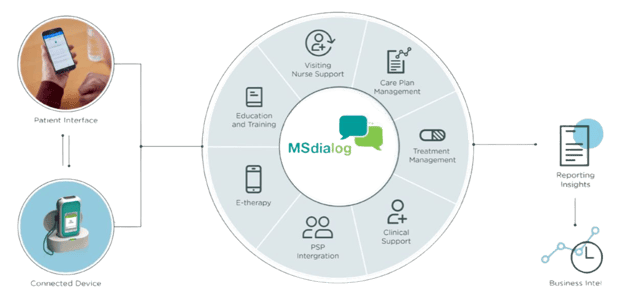

A connected injection device allows patients to self-inject and sends information wirelessly to each secure program server. The connected injection device records the date, time, and dosage of each injection so that an accurate dosing history can be discussed with a patient, allowing physicians to monitor and improve adherence to therapy.
The patient mobile app allows patients to provide feedback on their health and to track their own adherence and treatment progress. It also provides important economic and clinical real-world data, facilitating service planning and reconfiguration at country and regional levels.


Patients with RMS self-injecting REBISMART® had excellent adherence at 12 months, which was associated with good clinical outcomes
SMART Observational Study
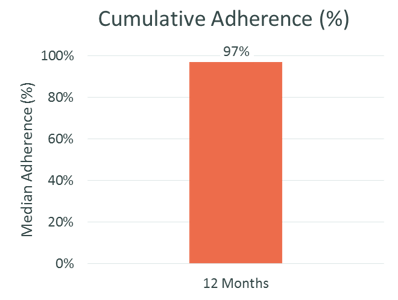
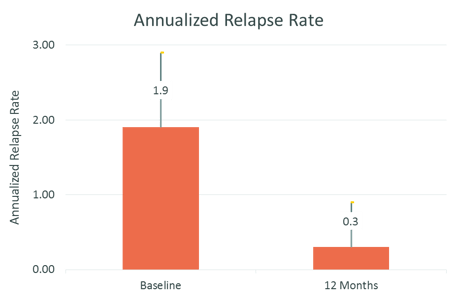
Treatment with REBISMART was efficacious: 80% of patients were relapse-free at 12 months, mean ARR was significantly lower at 12 months and EDSS did not increase during the study period
Source: Adherence to, and effectiveness of, subcutaneous interferon β-1a administered by RebiSmart® in patients with relapsing multiple sclerosis: results of the 1-year, observational SMART study: Expert Opinion on Drug Delivery, 2015, 12:8, 1239-1250
Effect of adherence on outcomes STAR Observational Study
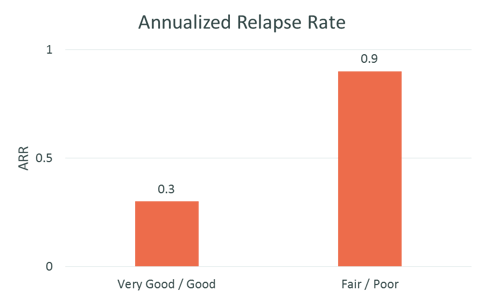
Treatment with REBISMART was efficacious: 80% of patients were relapse-free at 12 months, mean ARR was significantly lower at 12 months and EDSS did not increase during the study period
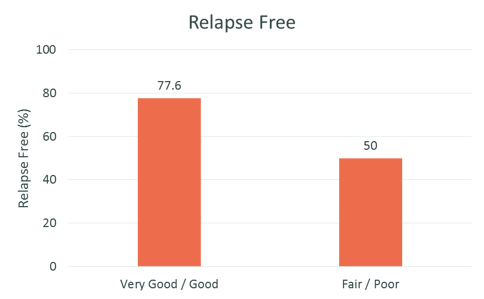
Source: The STAR Study: A Real-World, International, Observational Study of the Safety and Tolerability of, and Adherence to, Serum Free Subcutaneous Interferon β-1a in Patients With Relapsing Multiple Sclerosis; Hupperts, Raymond et al.; Clinical Therapeutics, Volume 36, Issue 12, 1946 – 1957
To learn more about how we help life science companies develop robust digital health strategies get in touch with us today
Contact UsSign up to our newsletter and marketing communications to be the first to receive our latest news, events and resources.
Copyright © 2016 - 2026 Silicon & Software Systems Ltd.